Protist infections in mountain gorillas across Virunga Massif – Bachelor of Science Carine Uwamahoro, BSc
Bachelor of Science Carine Uwamahoro, BSc
Master's thesis
Protist infections in mountain gorillas across Virunga Massif
Abstract:
Prepared by: Carine Uwamahoro (Dian Fossey Gorilla Fund) Research project for a master degree at Wildlife and Livestock Production, Management and Conservation at the Faculty of Tropical AgriSciences, the Czech University of Life Sciences Prague, Czech Republic Supervisors: Dr. Klara Petrzelkova (Institute of Vertebrate Biology, Czech Academy of Sciences) and prof. Martin Kvac (Institute of Parasitology, Biology Centre of Czech Academy of Sciences) Advisors: Dr. Winnie Eckard (Dian Fossey Gorilla Fund), Dr. Julius Nziza (Mountain Gorilla Veterinary Project) Introduction Parasites are known as organisms that can cause harm and diseases to the infected host. Giardia intestinalis, Cryptosporidium spp. and microsporidia of genera Encephalitozoon and Enterocytozoon are common intestinal unicellular parasites (protists) infecting humans and domesticated animals, including livestock, dogs and cats and they are spread by the fecal-oral route via environmentally resistant transmission stages (Didier et al., 1998; Mathis et al., 2005; Ryan et al., 2014; Thompson et al., 2008). These protists also infect great apes and other nonhuman primates (Graczyk et al., 2002; Salzer et al., 2007; Sak et al., 2011; 2013; 2014). In immunocompetent humans, the symptoms of Giardia and Cryptosporidium infections are usually mild and self-limiting, while in immunodeficient hosts they are characterized by chronic diarrhea, abdominal cramps, weight loss and can even cause death (Epe et al., 2010; Bouzid et al., 2013). Human cryptosporidiosis, microsporidiosis and giardiasis emerged as important opportunistic diseases when AIDS became pandemic. Thousands of HIV-infected patients with chronic diarrhea attributed to these organisms have been reported worldwide (Mathis et al., 2005). Microsporidial infections caused by Encephalitozoon spp. and E. bieneusi are characterized by a variety of pathologies ranging from being asymptomatic to lethal infections, mainly in immunodeficient hosts (Didier et al., 2005). Several studies showed high prevalence of asymptomatic microsporidia infection in captive and habituated great apes, while Cryptosporidium spp. and G. intestinalis seems to be less frequent (summarized in Modrý el al., 2018). Because Giardia, Cryptosporidium and microsporidia infections result from faecal-oral transmission through ingestion of contaminated water and food, transmission might occur between humans, domestic animals and wildlife including nonhuman primates sharing habitat (Sprong et al., 2009; Thompson & Monis, 2012). However, only genotyping and subtyping of particular isolates can provide the essential information about the epidemiology and zoonotic potential of these protists in nonhuman primates. Previous studies that sequenced phylogenetically informative genes of G. intestinalis, Cryptosporidium spp. and microsporidia in wild great apes confirmed potential transmission among humans, domestic animals and mountain gorillas (Gorilla beringei beringei) in Uganda (Nizeyi et al., 1999; Graczyk et al., 2001; Nizeyi et al., 2002a,b; Nolan et al., 2017) and Rwanda (Hogan et al., 2014; Sak et al., 2014), and western gorillas (Gorilla gorilla gorilla) in Central African Republic (Sak et al., 2013), and within orangutan populations (Pongo abelii and Pongo pygmaeus) on Sumatra (Mynářová et al., 2016). The mountain gorillas classified as an endangered subspecies with only two isolated populations remaining in Bwindi Impenetrable National Park, Uganda and the Virunga Massif (VM) at the borders of Rwanda, Uganda, and the Democratic Republic of Congo. The VM population declined in 1970s (Weber & Vedder, 1983), but the numbers stabilized in the 1980s, and since then the population has been increasing with annual growth rates around 3–4% between 2003–2016 (Gray et al., 2010; Granjon et al., 2020). However, this increase has been almost entirely attributed to the growth rate in habituated groups (Gray et al., 2010; Gray et al., 2013; Granjon …viacméněAbstract:
et al., 2020) due to the “extreme conservation” measures these groups benefit from, such as daily group monitoring and protection, veterinary interventions and anti-poaching patrols (Robbins et al., 2011). The population increase has not been uniform across the VM, which may be due to varying ecological conditions that are linked to different habitat types (Kalpers et al., 2003; Gray et al., 2010, 2013; Hickey et al., 2019). Moreover, the potential for spatial expansion is extremely limited due to dense human communities living nearby. Consequently, as gorilla population densities increase, their susceptibility to infectious diseases may also increase. Gastrointestinal problems (chronic gastritis) linked to helminth infections have been already observed in the VM and may pose a threat to these endangered animals, which led to a study examining helminths patterns across the VM and Bwindi Impenetrable NP. Striking geographic differences in strongylid infections (both intensity of infection and community composition) within the VM with higher egg counts and specific gastric strongylid taxa occurring mostly in areas with high occurrences of gastric disease (area between Karisimbi and Visoke) were revealed (Petrželková et al., 2021, Petrželková et al., unpublished data). This highly collaborative study pointed to new challenges emerging as possible “side effects” of the remarkable conservation success of the past few decades and calls for examining patterns of other parasite infections in VM gorillas. Unraveling the patterns of parasite infections in both gorilla populations, evaluating host exposure to infective parasite stages, and studying susceptibility to infection and its consequences on host health is an important next step for the survival of small, isolated populations of mountain gorillas. Only limited work has been done to explore the molecular diversity of gastrointestinal protists in VM mountain gorillas with exception of the studies by Sak et al. (2014) and Hogan et al. (2014), showing presence and a diversity of those important opportunistic protists in VM gorillas. Sak et al. (2014) detected various genotypes of Enterocytozoon bieneusi (genotype EbpA, D, C, gorilla 2 and five novel genotypes gorilla 4–8), Encephalitozoon cuniculi genotype I and II, Cryptosporidium muris and C. meleagridis. Although Sak et al. (2014) did not detect any Giardia infection, Hogan et al. (2014) found G. intestinalis in 9 of 130 mountain gorillas. Sequence analysis of the GDH Glutamate dehydrogenase gene (GDH) demonstrated the presence of G. intestinalis assemblage B in 6 out of 9 successfully sequenced samples but only in gorillas from the area between volcanos Karisimbi and Sabinyo. Importantly, Cryptosporidium sp. infections were identified only in groups ranging in forest areas including the volcanoes Karisimbi and Visoke, and the occurrence of E. cuniculi in groups ranging between Karisimbi and Visoke was significantly higher in comparison to other groups from other areas (Sak et al., 2014), whereas Giardia infections were missing in the groups ranging between the volcanoes Sabinyo and Muhavura (Hogan et al., 2014). However, only limited number of groups/samples were examined in these studies, and additionally, the mountain gorillas ranging between Karisimbi and Visoke have experienced major changes in the social structure after the samples for Sak et al. (2014) were collected. Three stable multi-male groups divided into 9-12 smaller groups including multiple one-male groups (Caillaud et al., 2014, 2020). Consequently, this threefold increase in group densities in this area led to an increase in group home range overlap, a reduction in home range areas used exclusively by a single group, and annual intergroup encounter rates tripled(Caillaud et al., 2020). Such small groups, which are often one-male groups, may experience higher stress levels than large groups, which are often multi-male groups, because they have higher home range overlaps with neighboring groups, inter-group encounter rates, and are more vulnerable to infanticide by external males. This all could affect the epidemiology of those parasitic protists and calls for a follow-up study examining patterns of those opportunistic pathogens in VM gorillas. Aims of the study The main goal of this study is to assess patterns of Cryptosporidium, Giardia and microsporidia infections in endangered mountain gorilla across the entire VM. Specific aims have been determined as follows: •Determine the molecular diversity of Cryptosporidium, Giardia and microsporidia infections in VM gorillas, and compare the results with the previous study (Sak et al., 2014) to assess possible temporal changes in these protist infections in last ~ 10 years •Assess geographical differences in Cryptosporidium, Giardia and microsporidia infections in gorillas within the VM •Evaluate the impact of host and environmental factors on these protist infections Material and methods Study site and animals The study will be carried out on samples collected across the entire range of the Virunga Massif mountain gorilla population (VM, 451 km2). VM is characterized by an altitude ranging from 2000 to 4500 meters above sea level (MASL) (McNeilage, 2001), with different vegetation types mainly determined by elevation, including the alpine zone (above 3,600 MASL), sub-alpine zone (3,200–3,600 MASL), Hagenia – Hypericum zone (2800–3200 MASL), lower altitude forest (mixed forest) (1,600–2,500 MASL), bamboo zone (2500–2800 MASL), disturbed woodland (2300–2800 MASL), open grassland and swamp (occurring in various altitudes) (Owiunji, 2005). We divide the VM into four areas according to volcano positions (modification of Weber & Vedder, 1983). This geographic division is used as a proxy, which reflects observed variation in the occurrence of gastrointestinal diseases, as well as differences in vegetation types, and in historical subpopulation growth and associated current social structure across the VM gorilla distribution range. The areas were namely: Karisimbi-Visoke (K_V), Visoke-Sabyinyo (V_S), Sabyinyo-Muhavura (S_M), Mikeno (M). Samples approximately 100 will be selected from a set of nest samples collected from monitored groups in 2018 during the helminth study (Petrželková et al., 2021, Petrželková et al., unpublished data) with a balanced number across the defined areas (K_V, V_S, S_M, M). Data about group size, and size of home range, gorilla density and exact vegetation within a home range will be obtained from Dian Fossey Gorilla Fund International and Mountain Gorilla Veterinary Project databases. DNA extraction, PCR amplification, sequencing and genotyping DNA isolation. Total genomic DNA will be extracted from 200 mg of feces by bead disruption for 60 s at 5.5 m/s using 0.5 mm glass beads in a FastPrep®24 Instrument (MP Biomedicals, CA, USA) followed by isolation/purification using a commercially available kit. Purified DNA will be stored at −20 °C or –80 °C prior to being used for PCR. Genotyping. A nested PCR approach will be used to amplify a partial region of the small subunit rRNA (18S rDNA) of Cryptosporidium spp. (Xiao et al., 1999, Jiang et al., 2005) with the modification described in Holubová et al. (2019), 18S rDNA and GDH of Giardia intestinalis (Hopkins et al., 1997; Read et al., 2004), ITS of Encephalitozoon spp. (Katzwinkel-Wladarch et al., 1996) and Enterocytozoon bieneusi (Buckholt et al., 2002). Samples tested positive for the presence of Cryptosporidium-specific 18S rDNA and do not match with known species/genotypes will be characterised at using nested PCR targeting COWP (Pedraza-Diaz et al., 2001), actin (Sulaiman et al., 2002), HSP70 (Morgan et al., 2001), gp60 (Alves et al., 2003) and/or TRAP-C1 (Spano et al., 1998) genes. Molecular grade water will used as a negative control. DNA of C. occultus or C. proliferans will be used as a positive control for detection of Cryptosporidium spp., Giardia muris for G. intestinalis, Encephalitozoon hellem 2A for Encephalitozoon spp. and E. bieneusi PtEbIX for E. bieneusi. DNA of C. hominis subtype family Ib will be used as a positive control for the amplification of the gp60 fragment. The secondary PCR products will be separated by electrophoresis on an agarose gel, stained with ethidium bromide and visualised under UV illumination. Sequencing. All amplicons will be purified using the GenElute™ Gel Extraction Kit (Sigma-Aldrich, St. Louis, MO, USA) or equivalent kit and directly sequenced using the secondary PCR primers at commercial companies (e.g., Eurofins, SeqMe, Macrogen). Phylogeny analysis Chromatogram analysis will be performed using software Chromas Pro 2.1.4 (Technelysium, Pty, Ltd., South Brisbane, Australia), and sequences will be verified by BLAST analysis (https://blast.ncbi.nlm.nih.gov/Blast.cgi). The sequences obtained in this study and reference sequences obtained from GenBank will be aligned using the software MAFFT version 7 online server (http://mafft.cbrc.jp/alignment/server/). The alignments will be manually trimmed and edited in BioEdit v.7.0.5 (Hall, 1999). Phylogenetic analysis will be performed using the maximum likelihood (ML) method, using evolutionary models selected by MEGAX (Kumar et al., 2018). Bootstrap supports will be calculated from 1000 replications. Phylogenetic trees will be produced by MEGAX and further edited for visualisation purposes with Corel DrawX7 (Corel Corporation, Ottawa, Ontario, Canada). Species-specific divergences will be identified from proportional distances (%) which will be calculated in the software program Geneious v11.0.3 (Kearse et al., 2012). Statistical Analyses Based on the number of detected Cryptosporidium, Giardia and microsporidia infections, most suitable statistical approach will be chosen. If only few infections will be detected (e.g. Giardia, Cryptosporidium), complex statistical analyses will not be conducted and prevalence of the infections among VM areas will be compared using Fisher exact test. In the case of a high prevalence of microsporidia infection, we run GLM to assess the impact of group size, and size of group home range, gorilla density and vegetation type within a group’s home range. All the analyses will be performed in software R 3.5.0 (R Core Team, 2020). Budget •DNA isolation – 450 USD •PCR for three pathogens – 1400 USD •sequencing – 330 USD •lab disposables – 220 USD References Alves, M., Xiao, L. H., Sulaiman, I., Lal, A. A., Matos, O. & Antunes, F. (2003). Subgenotype analysis of Cryptosporidium isolates from humans, cattle, and zoo ruminants in Portugal. Journal of Clinical Microbiology, 41, 2744–2747. Bouzid, M., Hunter, P. R., Chalmers, R. M. & Tyler, K. M. (2013). Cryptosporidium pathogenicity and virulence. Clinical Microbiology Reviews, 26, 115–134. Buckholt, M. A., Lee, J. H. & Tzipori, S. (2002). Prevalence of Enterocytozoon bieneusi in swine: an 18-month survey at a slaughterhouse in Massachusetts. Applied and Environmental Microbiology, 68, 2595–2599. Caillaud, D., et al. (2020). Violent encounters between social units hinder the growth of a high-density mountain gorilla population. Science Advances, 6, eaba0724. Caillaud, D., Ndagijimana, F., Giarrusso, A. J., Vecellio, V. & Stoinski, T. S. (2014). Mountain gorilla ranging patterns: Influence of group size and group dynamics. American Journal of Primatology, 76, 730–746. Cama, V. A. & Mathison, B. A. (2015). Infections by intestinal coccidia and Giardia duodenalis. Clinics in laboratory medicine, 35, 423–444. Didier, E. S., Snowden, K. F. & Shadduck, J. A. (1998). Biology of microsporidian species infecting mammals. Advances in Parasitology, 40, 283–320. Didier, E.S., Snowden, K.F., Shadduck, J.A. (1998). Biology of microsporidian species infecting mammals. Advanced Parasitology, 40, 283–320. Epe, C., Rehkter, G., Schnieder, T., Lorentzen, L. & Kreienbrock, L. (2010). Giardia in symptomatic dogs and cats in Europe: Results of a European study. Veterinary Parasitology, 173, 32–38. Graczyk, T., DaSilva, A., Cranfield, M., Nizeyi, J., Kalema, G. & Pieniazek, N. (2001). Cryptosporidium parvum Genotype 2 infections in free-ranging mountain gorillas (Gorilla gorilla beringei) of the Bwindi Impenetrable National Park, Uganda. Parasitology Research, 87, 368–370. Graczyk, T. K., Bosco-Nizeyi, J. B., Ssebide, B., Thompson, R. C. A., Read, C. & Cranfield, M. R. (2002). Anthropozoonotic Giardia duodenalis genotype (assemblage A) infections in habitats of free-ranging human-habituated gorillas, Uganda. Journal of Parasitology, 88, 905–909. Granjon, A.-C., et al. (2020). Estimating abundance and growth rates in a wild mountain gorilla population. Animal Conservation, 23, 455–465. Gray, M., et al. (2010). Virunga Massif Mountain Gorilla Census – 2010 Summary Report. GVTC, IGCP & partners, Kigali, Rwanda. Gray, M., et al. (2013). Genetic census reveals increased but uneven growth of a critically endangered mountain gorilla population. Biological Conservation, 158, 230–238. Hall, T. A. (1999). BioEdit: a user-friendly biological sequence alignment editor and analysis program for Windows 95/98/NT. Nucleic Acids Symposium Series, 41, 95–98. Hickey, J. R., Granjon, A.-C., Vigilant, L. et al. (2019). Virunga 2015–2016 surveys: Monitoring mountain gorillas, other select mammals, and illegal activities. GVTC, IGCP & partners, Kigali, Rwanda. Hill, D. E. & Dubey, J. P. (2018). Toxoplasma gondii as a parasite in food: analysis and control. In Thakur, S. & Kniel, K. E. (Eds.), Preharvest Food Safety (pp. 227–247). ASM Press, Washington, DC, USA. Hogan, J. N., et al. (2014). Giardia in mountain gorillas (Gorilla beringei beringei), forest buffalo (Syncerus caffer), and domestic cattle in volcanoes national park, Rwanda. Journal of Wildlife Diseases, 50, 21–30. Holubová, N., et al. (2019). Cryptosporidium proventriculi sp. n. (Apicomplexa: Cryptosporidiidae) in Psittaciformes birds. European Journal of Protistology, 69, 70–87. Hopkins, R. M., Meloni, B. P., Groth, D. M., Wetherall, J. D., Reynoldson, J. A. & Thompson, R. C. (1997). Ribosomal RNA sequencing reveals difference between the genes of Giardia isolates recovered from humans and dogs living in the same locality. Journal of Parasitology, 83, 44–51. Jiang, J., Alderisio, K. A. & Xiao, L. (2005). Distribution of Cryptosporidium genotypes in storm event water samples from three watersheds in New York. Applied and Environmental Microbiology, 71, 4446–4454. Kalpers, J. et al. (2003). Gorillas in the crossfire: population dynamics of the Virunga mountain gorillas over the past three decades. Oryx, 37, 326–337. Katzwinkel-Wladarsch, S., Lieb, M., Heise, W., Löscher, T., & Rinder, H. (1996). Direct amplification and species determination of microsporidian DNA from stool specimens. Tropical Medicine & International Health, 1, 373–378. Kearse, M., et al. (2012). Geneious Basic: An integrated and extendable desktop software platform for the organization and analysis of sequence data. Bioinformatics, 28, 1647–1649. Kumar, S., Stecher, G., Li, M., Knyaz, C., Tamura, K. (2018). MEGA X: Molecular Evolutionary Genetics Analysis across Computing Platforms. Molecular Biology and Evolution, 35, 1547–1549. Mathis, A., Weber, R. & Deplazes, P. (2005). Zoonotic potential of the microsporidia. Clinical Microbiology Reviews, 18, 423–445. Modry, D., Pafco, B., Petrzelkova, K. J., Hasegawa, H. (2018). Parasites of Apes, An Atlas of Coproscopic Diagnostics. Edition Chimaira, Frankfurt am Main. McNeilage, A. (2001). Diet and habitat use of two mountain gorilla groups in contrasting habitats in the Virunga. in Mountain gorillas: three decades of research at Karisoke. In Robbins, M. M., Sicotte, P. & Stewart, K. J. (Eds.), Mountain Gorillas (pp. 265–292). Cambridge University Press, Cambridge, UK. Morgan, U. M., et al. (2001). Molecular and phylogenetic characterisation of Cryptosporidium from birds. International Journal for Parasitology, 31, 289–296. Mynářová Foitová, I., et al. (2016). Prevalence of Cryptosporidium spp., Enterocytozoon bieneusi, Encephalitozoon spp. and Giardia intestinalis in wild, semi-wild and captive orangutans (Pongo abelli and Pongo pygmaeus) on Sumatra and Borneo, Indonesia. PLoS ONE, 11, e0152771. Nizeyi, J. B., Mwebe, R., Nanteza, A., Cranfield, M. R., Kalema, G. R. & Graczyk, T. K. (1999). Cryptosporidium sp. and Giardia sp. infections in mountain gorillas (Gorilla gorilla beringei) of the Bwindi Impenetrable National Park, Uganda. Journal of Parasitology, 85, 1084–1088. Nizeyi, J. B., Cranfield, M. R. & Graczyk, T. K. (2002a). Cattle near the Bwindi Impenetrable National Park, Uganda, as a reservoir of Cryptosporidium parvum and Giardia duodenalis for local community and free-ranging gorillas. Parasitology Research, 88, 380–385. Nizeyi, J. B., Sebunya, D., Dasilva, A. J., Cranfield, M. R., Pieniazek, N. J. & Graczyk, T. K. (2002b). Cryptosporidium in people sharing habitats with free-ranging mountain gorillas (Gorilla gorilla beringei), Uganda. The American Journal of Tropical Medicine and Hygiene, 66, 442–444. Nolan, M. J., et al. (2017). Molecular characterisation of protist parasites in human-habituated mountain gorillas (Gorilla beringei beringei), humans and livestock, from Bwindi impenetrable National Park, Uganda. Parasites & Vectors, 10, 340. Owiunji, I., et al. (2005). The biodiversity of the Virunga Volcanoes. Available at https://programs.wcs.org/portals/49/media/file/volcanoes_biodiv_survey.pdf. Parsons, M. B., et al. (2015). Epidemiology and molecular characterization of Cryptosporidium spp. in humans, wild primates, and domesticated animals in the Greater Gombe Ecosystem, Tanzania. PLoS Neglected Tropical Diseases, 9, e0003529. Pedraza-Diaz, S., Amar, C., Nichols, G. L. & McLauchlin, J. (2001). Nested polymerase chain reaction for amplification of the Cryptosporidium oocyst wall protein gene. Emerging Infectious Diseases, 7, 49–56. Petrzelkova, et al. (2021). Heterogeneity in patterns of helminth infections across populations of mountain gorillas (Gorilla beringei beringei). Scientific Reports, 11, 10869. Read, C. M., Monis, P. T. & Thompson, R. C. A. (2004). Discrimination of all genotypes of Giardia duodenalis at the glutamate dehydrogenase locus using PCR-RFLP. Infection, Genetics and Evolution, 4, 125–130. R Core Team (2020). R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. URL https://www.R-project.org/ Robbins, M. M., et al. (2011). Extreme conservation leads to recovery of the Virunga mountain gorillas. PLoS ONE, 6, e19788. Ryan, U., Fayer, R. & Xiao, L. (2014). Cryptosporidium species in humans and animals: current understanding and research needs. Parasitology, 141, 1667–1685. Salzer, J. S., Rwego, I. B., Goldberg, T. L., Kuhlenschmidt, M. S. & Gillespie, T. R. (2007). Giardia sp. and Cryptosporidium sp. infections in primates in fragmented and undisturbed forest in Western Uganda. Journal of Parasitology, 93, 439–440. Sak, B., et al. (2011). Diversity of microsporidia (Fungi: Microsporidia) among captive great apes in European zoos and African sanctuaries: evidence for zoonotic transmission? Folia Parasitologica, 58, 81–86. Sak, B., et al. (2013). Long-term monitoring of microsporidia, Cryptosporidium and Giardia infections in western Lowland Gorillas (Gorilla gorilla gorilla) at different stages of habituation in Dzanga Sangha Protected Areas, Central African Republic. PLoS ONE, 8, e71840. Sak, B., Petrželková, K. J., Květoňová, D., Mynářová, A., Pomajbíková, K., Modrý. D & Kváč, M. (2014). Diversity of microsporidia, Cryptosporidium and Giardia in mountain gorillas (Gorilla beringei beringei) in Volcanoes National Park, Rwanda. PloS ONE, 9, e109751. Spano, F., Putignani, L., Naitza, S., Puri, C., Wright, S. & Crisanti, A. (1998). Molecular cloning and expression analysis of a Cryptosporidium parvum gene encoding a new member of the thrombospondin family. Molecular and Biochemical Parasitology, 92, 147–162. Sprong, H., Cacciò, S.M., van der Giessen, J.W. (2009). Identification of zoonotic genotypes of Giardia duodenalis. PLoS Neglected Tropical Diseases, 3, e558. Sulaiman, I. M., Lal, A. A. & Xiao, L. (2002). Molecular phylogeny and evolutionary relationships of Cryptosporidium parasites at the actin locus. Journal of Parasitology, 88, 388–394. Thompson, R. C. A. & Monis, P. (2012). Giardia - From genome to proteome. Advances in Parasitology, 78, 57–95. Thompson, R. C. A., Palmer, C. S. & O’Handley, R. (2008). The public health and clinical significance of Giardia and Cryptosporidium in domestic animals. The Veterinary Journal, 177, 18–25. Weber, A. W. & Vedder, A. (1983). Population dynamics of the Virunga gorillas: 1959–1978. Biological Conservation, 26, 341–366. Xiao, L., et al. (1999). Genetic diversity within Cryptosporidium parvum and related Cryptosporidium species. Applied and Environmental Microbiology, 65, 3386–3391. …viacméně
Jazyk práce: English
Datum vytvoření / odevzdání či podání práce: 15. 8. 2022
Obhajoba závěrečné práce
- Vedúci: Klára Petrželková, Ph.D.
Citační záznam
Citace dle ISO 690:
UWAMAHORO, Carine. \textit{Protist infections in mountain gorillas across Virunga Massif}. Online. Diplomová práca. Praha: Česká zemědělská univerzita v Praze, Faculty of Tropical AgriSciences. 2022. Dostupné z: https://theses.cz/id/v8nvnj/.
UWAMAHORO, Carine. <i>Protist infections in mountain gorillas across Virunga Massif</i>. Online. Diplomová práca. Praha: Česká zemědělská univerzita v Praze, Faculty of Tropical AgriSciences. 2022. Dostupné z: https://theses.cz/id/v8nvnj/.
UWAMAHORO, Carine. Protist infections in mountain gorillas across Virunga Massif. Online. Diplomová práca. Praha: Česká zemědělská univerzita v Praze, Faculty of Tropical AgriSciences. 2022. Dostupné z: https://theses.cz/id/v8nvnj/.
@MastersThesis{Uwamahoro2022thesis,
AUTHOR = "Uwamahoro, Carine",
TITLE = "Protist infections in mountain gorillas across Virunga Massif [online]",
YEAR = "2022 [cit. 2024-10-20]",
TYPE = "Diplomová práca",
SCHOOL = "Česká zemědělská univerzita v Praze, Faculty of Tropical AgriSciencesPraha",
NOTE = "SUPERVISOR: Klára Petrželková, Ph.D.",
URL = "https://theses.cz/id/v8nvnj/",
}
AUTHOR = "Uwamahoro, Carine",
TITLE = "Protist infections in mountain gorillas across Virunga Massif [online]",
YEAR = "2022 [cit. 2024-10-20]",
TYPE = "Diplomová práca",
SCHOOL = "Česká zemědělská univerzita v Praze, Faculty of Tropical AgriSciencesPraha",
NOTE = "SUPERVISOR: Klára Petrželková, Ph.D.",
URL = "https://theses.cz/id/v8nvnj/",
}
@MastersThesis{Uwamahoro2022thesis,
AUTHOR = {Uwamahoro, Carine},
TITLE = {Protist infections in mountain gorillas across Virunga Massif},
YEAR = {2022},
TYPE = {Diplomová práca},
INSTITUTION = {Česká zemědělská univerzita v Praze, Faculty of Tropical AgriSciences},
LOCATION = {Praha},
SUPERVISOR = {Klára Petrželková, Ph.D.},
URL = {https://theses.cz/id/v8nvnj/},
URL_DATE = {2024-10-20},
}
AUTHOR = {Uwamahoro, Carine},
TITLE = {Protist infections in mountain gorillas across Virunga Massif},
YEAR = {2022},
TYPE = {Diplomová práca},
INSTITUTION = {Česká zemědělská univerzita v Praze, Faculty of Tropical AgriSciences},
LOCATION = {Praha},
SUPERVISOR = {Klára Petrželková, Ph.D.},
URL = {https://theses.cz/id/v8nvnj/},
URL_DATE = {2024-10-20},
}
{{Citace kvalifikační práce
| příjmení = Uwamahoro
| jméno = Carine
| instituce = Česká zemědělská univerzita v Praze, Faculty of Tropical AgriSciences
| titul = Protist infections in mountain gorillas across Virunga Massif
| url = https://theses.cz/id/v8nvnj/
| typ práce = Diplomová práca
| vedoucí = Klára Petrželková, Ph.D.
| rok = 2022
| počet stran =
| strany =
| citace = 2024-10-20
| poznámka =
| jazyk =
}}
Plný text práce
Obsah online archivu závěrečné práce
Zveřejněno v Theses:- světu
Jak jinak získat přístup k textu
Instituce archivující a zpřístupňující práci: Česká zemědělská univerzita v Praze, Fakulta tropického zemědělstvíCzech University of Life Sciences Prague
Faculty of Tropical AgriSciencesMaster programme / specializace:
Wildlife and Livestock Production, Management and Conservation / Wildlife Management and Conservation
Práce na příbuzné téma
Seznam prací, které mají shodná klíčová slova.
- Žádné práce na příbuzné téma.
Názov
Vložil
Vložené
Práva
Složky
Soubory
-
Co je jinak přidání souboru
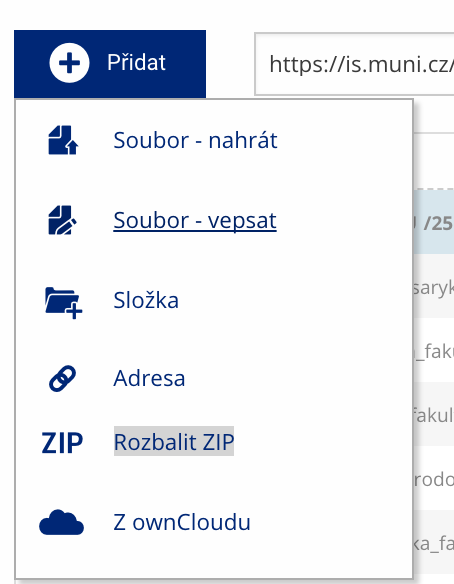 Soubor nebo složku lze nahrát pomocí tlačítka Přidat.
Soubor nebo složku lze nahrát pomocí tlačítka Přidat. -
Co je jinak další operace se soubory
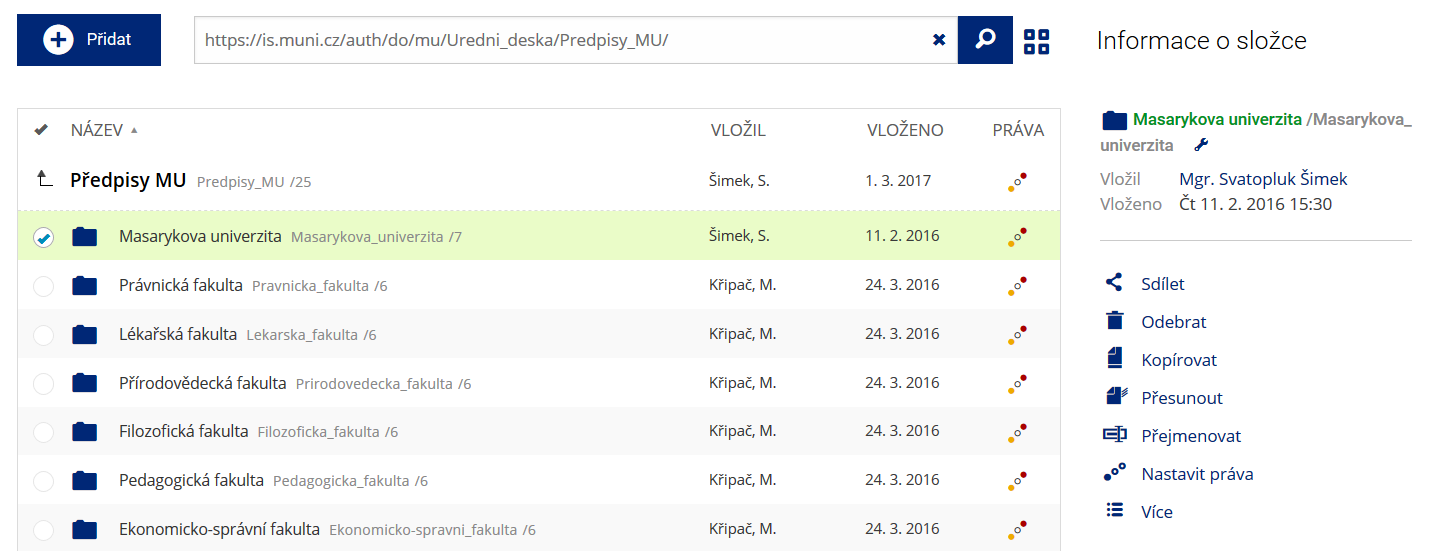 Podrobnosti lze zjistit označením příslušného řádku.
Podrobnosti lze zjistit označením příslušného řádku. -
Co je jinak pohled pro experty
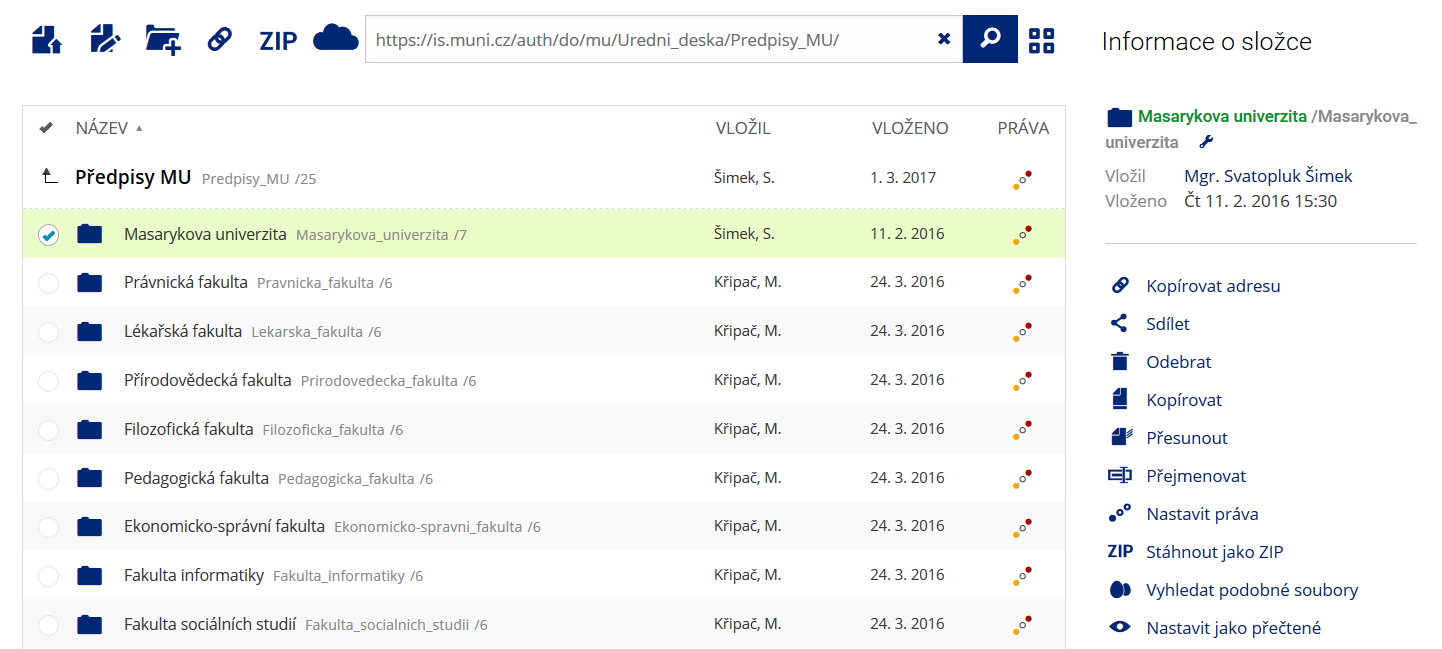 Pro častou práci je možné zvolit režim Více možností.
Pro častou práci je možné zvolit režim Více možností. -
Co je nové vyhledávání souborů
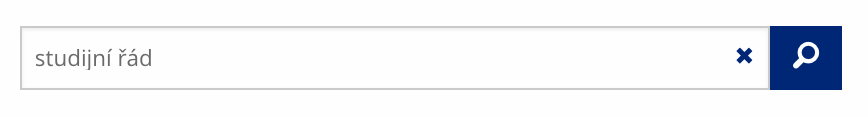 Vyhledávaný výraz můžete zadat přímo do adresního řádku.
Vyhledávaný výraz můžete zadat přímo do adresního řádku. -
Co je nové rychlý přístup k souborům
 Pomocí funkce Nedávné je možné se rychle vrátit k právě prohlíženým souborům. Oblíbené soubory je také možné označit Hvězdičkou.
Pomocí funkce Nedávné je možné se rychle vrátit k právě prohlíženým souborům. Oblíbené soubory je také možné označit Hvězdičkou. -
Co se chystá
 Připravujeme další vylepšení pro mobilní zařízení.
Připravujeme další vylepšení pro mobilní zařízení.